Aubagio Teriflunomide Injection, Sanofi Genzyme, 28 Film Coated Tablets, Rs 46872 /pack | ID: 21559365955

Sanofi launches 'once daily' oral tablet 'Aubagio®', for treatment of Multiple Sclerosis in India - The Pharma Times | Pharma & Health Care News Portal
SAŽETAK KARAKTERISTIKA LEKA 1. IME LEKA Aubagio®, 14 mg, film tablete INN: teriflunomid 2. KVALITATIVNI I KVANTITATIVNI SASTAV
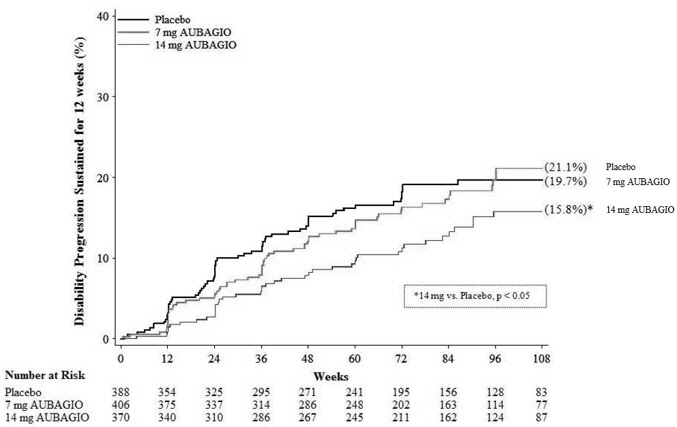
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AUBAGIO® safely and eff
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AUBAGIO® safely and eff

These highlights do not include all the information needed to use AUBAGIO® safely and effectively. See full prescribing information for AUBAGIO. AUBAGIO (teriflunomide) tablets, for oral use Initial U.S. Approval: 2012